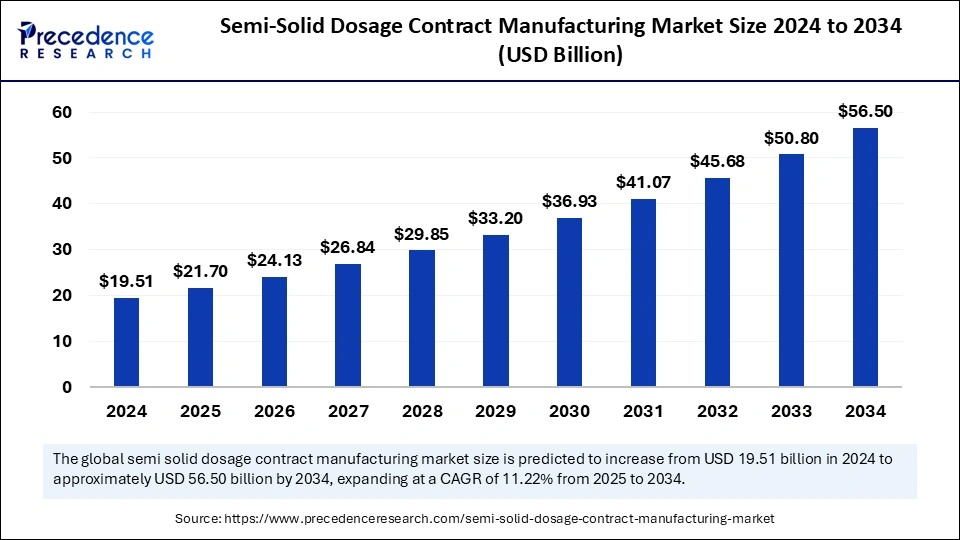
The global semi-solid dosage contract manufacturing market size was valued at USD 19.51 billion in 2024 and is expected to exceed around USD 56.50 billion by 2034, growing at a CAGR of 11.22%.
The semi-solid dosage contract manufacturing market is witnessing steady growth due to the rising demand for topical and transdermal drug delivery systems. Pharmaceutical companies are increasingly outsourcing manufacturing processes to specialized contract manufacturers to reduce costs, ensure regulatory compliance, and improve production efficiency. Semi-solid dosage forms, including creams, gels, ointments, and lotions, are widely used in dermatology, pain management, and hormone therapy, further driving market expansion.
Semi-Solid Dosage Contract Manufacturing Market Key Highlights
-
Asia Pacific accounted for more than 33% of the total market share in 2024.
-
North America is forecasted to expand at the highest CAGR of 10.67% in the coming years.
-
The topical segment remained the dominant type, contributing 50% of the market in 2024.
-
The transdermal segment is expected to register remarkable growth over time.
-
Among products, creams generated the largest revenue share in 2024.
-
Large-sized companies retained the leading position in the global market in 2024.
-
Medium and small-sized companies are likely to experience steady market expansion.
-
Pharmaceutical companies captured the highest share in the end-use segment in 2024.
-
Cosmeceutical companies are projected to grow at a promising rate in the forecast period.
Role of AI in the Semi-Solid Dosage Contract Manufacturing Market
Artificial Intelligence (AI) is revolutionizing the semi-solid dosage contract manufacturing market by enhancing production efficiency, quality control, and formulation development. AI-powered predictive analytics help contract manufacturers optimize production processes by identifying potential bottlenecks, minimizing waste, and ensuring consistent product quality.
Machine learning algorithms analyze vast datasets to improve formulation stability, enabling precise ingredient mixing and reducing variability in semi-solid drug formulations like creams, gels, and ointments. These advancements lead to better product consistency and compliance with regulatory standards.
AI also plays a crucial role in automating quality assurance and regulatory compliance. Computer vision and deep learning technologies can detect even the slightest inconsistencies in texture, viscosity, and ingredient dispersion, ensuring uniformity across batches. Additionally, AI-driven automation streamlines supply chain management by predicting demand fluctuations, optimizing inventory, and reducing production downtime.
With AI-driven digital twins and real-time monitoring systems, manufacturers can simulate different formulation scenarios and enhance process efficiency. As AI adoption continues to grow, it is expected to drive further innovation, reduce operational costs, and improve scalability in semi-solid dosage contract manufacturing.
Role of AI in Semi-Solid Dosage Contract Manufacturing Market
Artificial Intelligence (AI) is playing a transformative role in the semi-solid dosage contract manufacturing market by optimizing production processes, enhancing quality control, and improving regulatory compliance. AI-driven predictive analytics help manufacturers anticipate potential formulation issues, reduce batch failures, and optimize ingredient compositions.
Machine learning algorithms assist in streamlining drug formulation development by analyzing vast datasets to identify the most stable and effective formulations. Additionally, AI-powered automation in manufacturing facilities enhances precision and efficiency, minimizing human errors and reducing production costs.
AI is also revolutionizing quality assurance and regulatory compliance in contract manufacturing. AI-driven image recognition and real-time monitoring systems help detect inconsistencies in semi-solid formulations, ensuring product uniformity and safety. Natural Language Processing (NLP) tools assist in analyzing regulatory guidelines and automating compliance documentation, reducing the risk of non-compliance.
Furthermore, AI-integrated smart sensors enable real-time tracking of production parameters, ensuring consistency in temperature, viscosity, and other critical quality attributes. As AI technology advances, it is expected to further enhance innovation, efficiency, and scalability in semi-solid dosage contract manufacturing.
What is Semi-Solid Dosage Contract Manufacturing?
Semi-solid dosage contract manufacturing refers to the outsourcing of production for pharmaceutical and cosmeceutical products in semi-solid forms, such as creams, gels, ointments, pastes, and lotions. These dosage forms are commonly used for dermatological, transdermal, and mucosal drug delivery applications. Pharmaceutical companies often partner with contract manufacturers to leverage their expertise, reduce costs, and ensure compliance with regulatory standards.
Market Growth and Demand
The demand for semi-solid dosage forms is growing due to the increasing prevalence of skin-related diseases, the rising need for transdermal drug delivery, and advancements in formulation technologies. Contract manufacturers play a crucial role in helping pharmaceutical companies meet market demands while focusing on research and development. The market is expanding as companies seek efficient and cost-effective solutions for product development, testing, and large-scale manufacturing.
Key Services Offered
Contract manufacturers provide a wide range of services, including:
-
Formulation Development: Optimizing the composition of semi-solid drugs for stability and efficacy.
-
Analytical Testing: Ensuring product quality, safety, and compliance with regulatory standards.
-
Manufacturing and Packaging: Large-scale production with high precision and compliance with Good Manufacturing Practices (GMP).
-
Regulatory Support: Assisting pharmaceutical companies in meeting FDA, EMA, and other global regulatory requirements.
Technological Advancements
AI, automation, and advanced formulation technologies are revolutionizing semi-solid dosage contract manufacturing. AI-driven predictive analytics help optimize formulations, while automation enhances precision and reduces human errors. Innovations such as nanoemulsions, lipid-based drug delivery, and controlled-release formulations are improving the efficiency and effectiveness of semi-solid drugs.
Regulatory Considerations
The industry is highly regulated, with strict guidelines from agencies like the U.S. FDA, the European Medicines Agency (EMA), and other regulatory bodies worldwide. Compliance with GMP, stability testing, and quality assurance protocols is essential for contract manufacturers to ensure product safety and efficacy.
Semi-Solid Dosage Contract Manufacturing Market Scope
| Report Coverage | Details |
| Market Size by 2034 | USD 56.50 Billion |
| Market Size in 2025 | USD 21.70 Billion |
| Market Size in 2024 | USD 19.51 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 11.22% |
| Dominated Region | Asia Pacific |
| Fastest Growing Market | North America |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Type, Product, Company Size, End-use, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Semi-Solid Dosage Contract Manufacturing Market Dynamics
Market Drivers
Key factors propelling the market include increasing demand for dermatological and transdermal treatments, growing preference for outsourcing by pharmaceutical firms, and advancements in formulation technologies. Stringent regulatory requirements for drug manufacturing also encourage companies to partner with contract manufacturers that have the expertise and facilities to meet industry standards.
Market Opportunities
The rise in chronic skin conditions, growing geriatric population, and expanding pharmaceutical pipelines offer significant growth opportunities. Additionally, technological innovations in drug delivery, such as nanoemulsions and lipid-based formulations, are driving new developments in semi-solid dosage forms. Emerging markets with evolving healthcare systems also present lucrative opportunities for contract manufacturers.
Market Challenges
Challenges in the market include high initial investment costs for setting up advanced manufacturing facilities, strict regulatory compliance requirements, and quality control concerns. The complexity of scaling up semi-solid formulations without affecting product consistency can also pose difficulties for manufacturers. Supply chain disruptions and raw material shortages further add to industry challenges.
Regional Insights
- Assisted Living Market Size to Attain USD 8.60 Bn by 2034 - April 4, 2025
- Steam Education Market Size to Attain USD 52.74 Bn by 2034 - April 4, 2025
- Marine Sensors Market Size to Attain USD 59.45 Bn by 2034 - April 4, 2025

